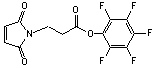
a-Maleimidoacetic acid-NHS
[55750-61-3]; C10H8N2O6; Mr=252.18; mp 174-175 °C
Spacer Arm length: 4.4 Ĺ.
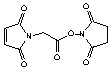
● Ultra short, sulfhydryl and amino reactive heterobifunctional
crosslinking reagent.1,2
1. May, J. M. (1989) Biochemistry 28, 1718.
2. Sayre, L. M., et.al. (1984) J. Med. Chem. 27, 1325.
Storage 0-4 °C.
[146474-00-2]; C6H8N2O2·CF3CO2H; Mr=254.17; mp 124 °C
●
Water soluble!
●
Derivatize Carboxy terminus or glutamic
acid side chains of peptides, to maleimido-peptides with EDC·HCl.
●
Introduce maleimide group into glycoproteins via Nakane1
coupling.
●
Used to interchange functional
groups.
1. Nakane, P.K. (1975) Ann. N.Y. Acad. Sci. 254, 203.
Storage 0-4 °C.
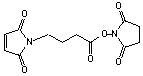
3-Maleimidopropionic acid NHS
[55750-62-4]; C11H10N2O6; Mr=266.21; mp 171 °C
Spacer Arm length: 6.9 Ĺ.
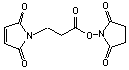
●
Short, sulfhydryl and amino reactive heterobifunctional crosslinking
reagent.1,2
● Used to
crosslink haptens to enzymes.3
●
Wide application in immunodiagnostics.4
1. Kitagawa, T. (1981) Enz. Immunoassay,
81.
2. Tanimori, H., et.al. (1983) J. Immunol.
Meth. 62, 123.
3. Hashida, S., et.al. (1984) J. Appl.
Biochem. 6, 56.
4. Moroder, L., et.al. (1983)
Biopolymers 22, 481.
Storage 0-4 °C.
1.0g
$425.00
C13H6NO4F5; Mr=335.19; mp 133-134 °C
Spacer Arm length: 6.9 Ĺ.
●
Heterobifunctional crosslinking reagent.
● Highly electrophilic
leaving group enables direct labeling of cytosine.
Storage 0-4 °C.
4-Maleimidobutyric acid NHS
[80307-12-6]; C12H12N2O6; Mr=280.24; mp 124-125 °C
Spacer Arm length: 6.8 Ĺ.
●
Sulfhydryl and amino reactive heterobifunctional crosslinking reagent.1,2
●
Introduces maleimide groups into IgM.3
●
Used to prepare enzyme-hapen conjugates.4
1. Keller, O., Rudinger, Z.J. (1975) Helv. Chim. Acta.
58, 531.
2. Tanimori, H., et.al. (1983) J. Immunol.
Meth. 62, 123.
3. Fujiwara, K., et.al. (1988) J. Immunol. Meth.
112, 77-83.
4. Moroder, L., et.al. (1983)
Biopolymers 22, 481.
Storage 0-4 °C.
250mg
1.0g
$235.00
$875.00
6-Maleimidocaproic acid NHS, 99%
[55750-63-5]; C14H16N2O6; Mr=308.3; mp 68-69 °C
Spacer Arm length: 9.4 Ĺ.
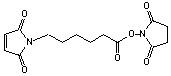
●
Sulfhydryl and amino reactive heterobifunctional crosslinking reagent.1
●
Reagent useful for crosslinking to proteins.2
●
Used to prepare site-specific glycosylation reagents for
peptides and proteins.3
1. Tanimori, H., et.al. (1981) J. Pharm. Dyn., 4, 812.
2. Partis, M.D., et.al. (1983) J.
Prot. Chem. 2, 263.
3. Ni, J.; Singh, S.; Wang, L.X. (2003)
Bioconjugate Chem. 14, 232-238.
Storage 0-4 °C.
500mg
$575.00
6-[N-(6-Maleimidocaproyl)]caproic acid NHS
C20H27N3O7; Mr=421.45
Spacer Arm length: 16.8 Ĺ.
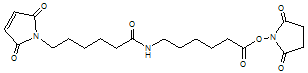
●
A C-6 chain extended analogue of EMCS.
●
Crosslinks amino and sulfhydryl groups.
Storage 0-4 °C.
6-Maleimidocaproic acid sulfo-NHS
C14H15N2O9SNa; Mr=410.33
Spacer Arm length: 9.4 Ĺ.
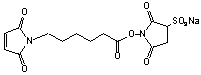
●
Water soluble!
●
Sulfhydryl and amino reactive heterobifunctional crosslinking reagent.1
●
Used to conjugate antisense morpholino oligomers to peptides.2
1. Kitagawa, T., et.al. (1981) Chem. Pharm. Bull. 29,
1130.
2. Moulton, H. M., Nelson, M. H.,
Hatlevig, S. A., Reddy, M. T., Iverson, P. L. (2004) Bioconjugate Chem.
15, 290-299.
Storage 0-4 °C.
1.0g
$1,000.00
6-Maleimidocaproic acid (2-nitro-4-sulfo) phenyl ester sodium salt
C16H15N2O9SNa; Mr=434.35
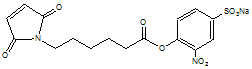
●
Water soluble!
●
Quantitate reaction with amines by release of 2-nitro 4-sulfo
phenol.1
●
Used to prepare a PEG-maleimide for pegylation of IL-2.2
1. Aldwin, L., Nitecki, D. (1987) Anal. Biochem. 164,
494.
2. Goodson, R. J., Katre, N. V. (1990)
Bio/Technology 8, 343-346.
Storage 0-4 °C.
1.0g
$725.00
11-Maleimidoundecanoic acid sulfo-NHS
C19H25N2O9SNa; Mr=485.47; mp 245 °C (Decomp)
Spacer Arm length: 15.7 Ĺ.
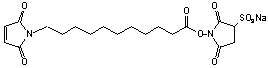
●
Water soluble!
Solubility: 1 mg/ 50mL
water.
●
Sulfhydryl and amino reactive heterobifunctional crosslinking reagent.
●
Used to configure bioluminescent enzyme immunoassays (BLEIA).1
1. Seto, Y.; Abe, K.; Itoh, M.; Sawai, H. (2002) Bioconjugate Chem. 13, 303-308.
Storage 0-4 °C.
Succinimidyl 6-[(3-maleimido)propionamido]-hexanoate
C17H21N3O7; Mr=379.33; mp 109-111 °C
Spacer Arm length: 14.3 Ĺ.
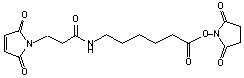
● Sulfhydryl and amino reactive heterobifunctional crosslinking reagent.
Storage 0-4 °C.
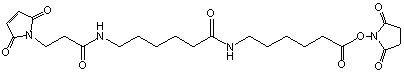
C23H32N4O8; Mr=492.52; mp 127-128.4 °C
Spacer Arm length: 18.8 Ĺ.
●
Sulfhydryl and amino reactive.
●
Heterobifunctional crosslinking reagent with extended chain
length of 20 atoms!
Storage 0-4 °C.
b-Maleimidopropionic acid hydrazide
C7H10N3O3·CF3CO2H; Mr=297.19; mp 116-117 °C
Spacer Arm length: 8.1 Ĺ.
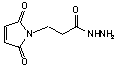
●
Sulfhydryl and carbonyl reactive heterobifunctional crosslinking
reagent.1
●
Used to prepare thiol reactive, luminescent metal chelates.2
1. Kitagawa, T., et.al. (1981) Chem. Pharm. Bull.
29, 1130.
2. Ge, P.; Selvin, P.R. (2003) Bioconjugate Chem.
14, 870-876.
Storage 0-4 °C.
e-Maleimidocaproic acid hydrazide
C10H15N3O3·CF3CO2H; Mr=339.27; mp 104-105 °C
Spacer Arm length: 11.8 Ĺ.
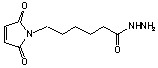
●
Sulfhydryl and carbonyl reactive heterobifunctional crosslinking
reagent.
●
Useful for preparing immunoconjugates of doxorubicin.1
1. Trail, P.A., et.al. (1993) Science, 261, 212-215.
Storage 0-4 °C.
11-Maleimidoundecanoic acid hydrazide
C15H25N3O3; Mr=295.38; mp 75 °C
Spacer Arm length: 19.0 Ĺ.
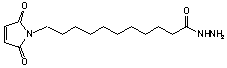
●
Sulfhydryl and carbonyl reactive heterobifunctional crosslinking
reagent.
●
Introduces maleimide group into glycoproteins.
●
Used to conjugate ligands to MoAb.1
1. Barth, R.F., et.al. (2004) Bioconjugate Chem. 15, 185-194.
Storage 0-4 °C.
[7423-55-4]; C7H7NO4; Mr=169.13; mp 105-107 °C
Spacer Arm length: 5.9 Ĺ.
●
Sulfhydryl reactive heterobifunctional crosslinking reagent.1
●
For preparing peptide-protein conjugates.2
●
Used to prepare peptide tracers and peptide-protein
conjugates.3
●
Useful in targeted protein functionalization.4
1. Rich, D. H., Gesellchen, P. D.,
Tong, A., Cheung, A., Buckner, C. K. (1975) J. Med. Chem. 18, 1004-1010.
2. Moroder, L., et.al. (1983) BioPolymers 22, 481.
3. Moroder, L., et.al. (1987) Hoppe-Seyler
368, 855.
4. Meredith, G. D., Wu, H. Y., Allbritton,
N. L. (2004) Bioconjugate Chem. 15, 969-982.
Storage 0-4 °C.
4-Maleimidobutyric acid
[57078-98-5]; C8H9NO4; Mr=183.2; mp 97-98 °C
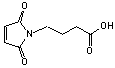
●
Heterobifunctional crosslinking reagent.
●
Modification reagent for thiol groups in proteins.1,2
●
Used to conjugate bleomycin.3
1. Rich, D. H., Gesellchen, P. D., Tong,
A., Cheung, A., Buckner, C. K. (1975) J. Med. Chem. 18, 1004-1010.
2. Griffiths, D.G. et.al. (1981) FEBS
Lett. 134, 261.
3. Fujiwara, K., Ysuna, M., (1981)
Cancer Res. 41, 4121.
Storage 0-4 °C.
6-Maleimidocaproic acid
[55750-53-3]; C10H13NO4; Mr=211.21; mp 88-90 °C
Spacer Arm length: 9.4 Ĺ.
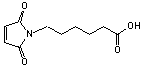
●
Sulfhydryl reactive heterobifunctional crosslinking reagent.
●
Widely used probe for introducing maleimide groups into biomolecules.
●
Probe for thiol groups in proteins.1,2
1. Griffith, D.G., et.al. (1981) FEBS Lett. 134, 261.
2. Keller, O., Rudinger, Z.J. (1975) Helv. Chim. Acta.
58, 531.
Storage 0-4 °C.
C15H23NO4; Mr=281.35; mp 89-90 °C
Spacer Arm length: 15.7 Ĺ.
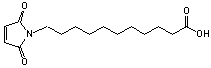
●
Sulfhydryl reactive heterobifunctional crosslinking reagent.1
●
For preparing peptide-protein conjugates.2
1. Rich, D. H., Gesellchen, P. D.,
Tong, A., Cheung, A., Buckner, C. K. (1975) J. Med. Chem. 18, 1004-1010.
2. Moroder, L., et.al. (1983) BioPolymers 22, 481.
Storage 0-4 °C.
[64987-85-5]; C16H18N2O6; Mr=334.33; mp 179 °C
Spacer Arm length: 11.6 Ĺ.
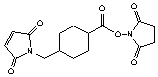
●
Sulfhydryl reactive heterobifunctional crosslinking reagent.1,2
●
Cyclohexane ring stabilizes maleimide group.
●
(Stable in 0.1 M phosphate, pH 7.0, 4 °C, 64 h.)2
●
Conjugating anti-digoxin F(ab´)2 fragments to b-galactosidase.3
●
Conjugating hIgG to alkaline phosphatase.4
●
Preparing immunogens.5
1. Maham, D.G., et.al. (1987) Anal. Biochem. 162, 163-170.
2. Yoshitake, S., et.al. (1979) Eur. J. Biochem. 101, 395-399.
3. Freytag, J.W., et.al. (1984) Clin. Chem. 30(9), 1494-1498.
4. Mahan, D. E., et.al. (1987) Anal. Biochem. 162, 163-170.
5. Peeters, J.M., et.al. (1989) J. Immunol. Meth. 120, 133-143.
Storage 0-4 °C.
250mg
1.0g
$137.50
$395.00
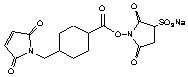
[92921-24-9]; C16H17N2O9SNa; Mr=436.37
Spacer Arm length: 11.6 Ĺ.
●
Sulfhydryl reactive heterobifunctional crosslinking reagent.1,2
●
Cyclohexane ring stabilizes maleimide group.
●
(Stable in 0.1 M phosphate, pH 7.0, 4 °C, 64 h.)2
●
Conjugating anti-digoxin Fab´ fragments to HRP via the hinge region.3
●
Preparation of enzyme-antibody conjugates.4
●
Water soluble!
1. Maham, D.G., et.al. (1987) Anal. Biochem. 162, 163-170.
2. Yoshitake, S., et.al. (1979) Eur. J. Biochem. 101, 395-399.
3. Hashida, S., et.al. (1984) J. Applied Biochem. 6, 56-63.
4. Samoszuk, M.K., et.al. (1989) Antibody Immunocon. Radiopharm.
2(1), 37-46.
Storage 0-4 °C.
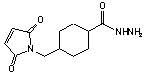
C12H17N3O3·CF3CO2H; Mr=365.31; mp >270 °C
●
Sulfhydryl and carbonyl reactive heterobifunctional crosslinking
reagent.
●
Glycoprotein reactive.
● Hydrazide group reacts with aldehydes to form stable hydrazone linkage.1
●
Cyclohexane ring stabilizes maleimide group.
●
(Stable in 0.1 M phosphate, pH 7.0, 4 °C, 64 h.)2
1. Chamow, S. M., et.al. (1992) J. Biol. Chem.
267(22), 15916-15922.
2. Yoshitake, S., et.al. (1979) Eur. J. Biochem. 101,
395-399.
Storage 0-4 °C.
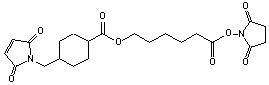
C22H29N3O7; Mr=447.44; mp 168-169 °C
Spacer Arm length: 16.1 Ĺ.
●
Sulfhydryl and carbonyl reactive heterobifunctional crosslinking
reagent.
●
Extended derivative of SMCC.
●
Cyclohexane ring stabilizes maleimide group.
●
(Stable in 0.1 M phosphate, pH 7.0, 4 °C, 64 h.)1
1. Yoshitake, S., et.al. (1979) Eur. J. Biochem.
101, 395-399.
Storage 0-4 °C.
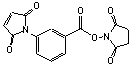
m-Maleimidobenzoyl N-hydroxysuccinimide
[58626-38-3]; C15H10N2O6; Mr=314.25; mp 175-177 °C
Spacer Arm length: 9.9 Ĺ.
●
Sulfhydryl and amino reactive heterobifunctional protein crosslinking
reagent.1
1. Kitagawa, T., et.al. (1981) Chem. Pharm. Bull. 29,
1130.
Storage 0-4 °C.
1.0g
$225.00
C15H9N2O9SNa; Mr=416.13
Spacer Arm length: 9.9 Ĺ.
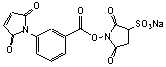
●
Water soluble
analog of MBS-1.1
1. Aithal, H.N., et.al. (1988) J. Immunol. Meth. 112,
63-70.
Storage 0-4 °C.
p-Maleimidobenzoyl N-hydroxysuccinimide
C15H10N2O6; Mr=314.25; mp 197-198 °C
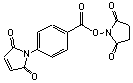
● Sulfhydryl and amino reactive heterobifunctional protein crosslinking reagent.
Storage 0-4 °C.
250mg
$82.50
(SVSB)
Succinimidyl-(4-vinylsulfonyl)benzoate
C13H11NSO6; Mr=309.29
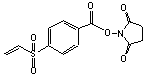
●
Amino and sulfhydryl reactive heterobifunctional
crosslinker.
● Hydrolytically stable vinylsulfone moiety!1,2
● Unlike maleimides, michael addition to vinylsulfone does not produce stereoisomers.
1. Morpurgo, M., et.al. (1996) Bioconjugate Chem. 7, 363-368.
2. Masri, M.S., et.al. (1988) J. Protein Chem. 7, 49-54.
Storage 0-4 °C.
N-succinimidyl-(4-iodoacetyl) aminobenzoate
C13H11N2O5I; Mr=402.15; mp 199-200 °C
Spacer Arm length: 10.6 Ĺ.
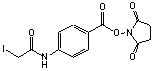
●
Sulfhydryl and amino reactive heterobifunctional protein crosslinking
reagent.
●
Conjugating antibodies to b-galactosidase.1
● Used to prepare
antibody-toxin conjugates.2
1. Weltman, J.K., et.al. (1983) Biotechniques 1,
148-152.
2. Cumber, A. J., et. al. (1985)
Methods in Enzymology Vol. 112, pp. 207-224.
Storage 0-4 °C.
C13H10N2O8INa; Mr=504.20
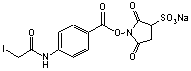
●
Sulfhydryl and amino reactive heterobifunctional protein crosslinking
reagent.
●
Water soluble
analog of SIAB.
●
Conjugating antibodies to b-galactosidase.1
1. Weltman, J.K., et.al. (1983) Biotechniques 1,
148-152.
Storage 0-4 °C.
p-Maleimidophenylisocyanate
C11H6N2O3; Mr=214.18; mp 121-123 °C
Spacer Arm length: 8.7 Ĺ.
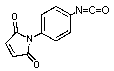
●
Sulfhydryl and hydroxyl reactive, heterobifunctional protein crosslinking
reagent.
●
For conjugation of b-estradiol, digitoxigenin,
and vitamin B12 hydroxyl.1
1. Palumbo, P.S., et.al. (1993) Bioconjugate Chem. 4,
212-218.
Storage 0-4 °C.
N-succinimidyl S-acetylthioacetate
[76931-93-6]; C8H9NO5S; Mr=231.22; mp 85-86 °C
Spacer Arm length: 2.8 Ĺ.
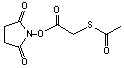
●
Reacts with primary amines to add protected sulfhydryls.1
●
Thioacetyl group can be deprotected with 0.02 M hydroxylamine hydrochloride
to render free sulfhydryl.
●
Used to add thiol groups to rabbit polyclonal IgG.2
●
Used to add a thiol group to Int 8.3
1. Duncan, R.J.S., et.al. (1983) Anal. Biochem.
132. 68-73.
2. Hermanson, G. T. (1996) Bioconjugate Techniques, Academic
Press, p.62.
3. Johnson, J.E., et. al. (2004) Bioconjugate Chem. 15,
807-813.
Storage 0-4 °C.
250mg
$137.50
N-succinimidyl S-acetylthiopropionate
[84271-78-3]; C9H11NO5S; Mr=245.24; mp 63-64 °C
Spacer Arm length: 4.1 Ĺ.
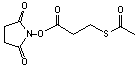
●
Reacts with primary amines to add protected sulfhydryls.1
●
Extended analog of SATA.
●
Thioacetyl group can be deprotected with 0.02 M hydroxylamine hydrochloride
to render free sulfhydryl.2
1. Duncan, R.J.S., et.al. (1983) Anal. Biochem. 132.
68-73.
2. Fuji, N., Akaji, K., Hayashi, Y., Yajima, H. (1985) Chem. Pharm. Bull. 33, 362-367.
Storage 0-4 °C.
Succinimidyl 3-(bromoacetamido)propionate
C9H11N2O5Br; Mr=308.19; mp 107-110.5 °C
Spacer Arm length: 6.2 Ĺ.
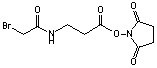
●
Sulfhydryl and amino reactive heterobifunctional crosslinking reagent.
●
Useful for preparing cyclic peptides, peptide conjugates, and polymers.1
1. Inman, J.K., et.al. (1991) Bioconjugate Chem. 2,
458-463.
Storage 0-4 °.
Succinimidyl iodoacetate
[39028-27-8]; C6H6NO4I; Mr=283.02; mp 148 °C
Spacer Arm length: 1.5 Ĺ.
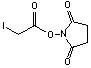
●
Sulfhydryl and amino reactive heterobifunctional protein
crosslinking reagent.1,2
●
Used to functionalize nanoparticles for application as
MR contrast agents.3
1. Eur. J. Biochem. (1984) 140, 63.
2. Rector, E.S., Schwenk, R.J., Tse, K.S., Sehon, A., Chan, H. (1978) J. Immun. Meth. 24, 321.
2. Wunderbaldinger, P.; Josephson, L.; Weissleder, R. (2002)
Bioconjugate Chem. 13, 264-268.
Storage 0-4 °C.
Succinimidyl bromoacetate
[42014-51-7]; C6H6NO4Br; Mr=237.00; mp 115-116 °C
Spacer Arm length: 1.5 Ĺ.
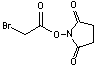
●
Sulfhydryl and amino reactive heterobifunctional
crosslinking reagent.1
● Sulfhydryl selectivity at neutral or slightly alkaline pH is comparable to iodoacetamide,
and perhaps greater than maleimide.2
●
Useful in the preparation of thiol reactive,
luminescent, lanthanide chelates.3
1. Cuatrecasas, P., et.al. (1969) J. Biol.
Chem. 244, 4316.
2. Bernatowicz, M.S., et.al. (1986) Anal. Biochem. 155, 95.
3. Ge, P., Selvin, P. R. (2004) Bioconjugate Chem. 15,
1088-1094.
Storage 0-4 °C.
Succinimidyl 4-(p-maleimidophenyl)butyrate
[79886-55-8]; C18H16N2O6; Mr=356.32; mp 130-131 °C
Spacer Arm length: 14.5 Ĺ.
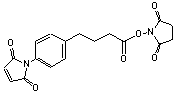
●
Sulfhydryl and amino reactive heterobifunctional
crosslinking reagent.1
●
Crosslinking to alkaline phosphatase.2
1. Martin, F. J., et.al. (1982) J. Biol. Chem. 257,
286-288.
2. Teale, J.M., et.al. (1986) J. Mol. Cell Immunol. 2,
283-292.
Storage 0-4 °C.
Succinimidyl-p-formyl-benzoate
[60444-78-2]; C12H9NO5; Mr=247.19; mp 163 °C
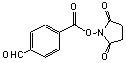
●
Amino reactive heterobifunctional crosslinking reagent.1,2
● Useful derivative for conjugating angiotensin to its receptor.3
1. Kraehenbuhl, J.P., et.al. (1974) J. Exptl. Med. 139, 208.
2. Tijssen, P., et.al. (1974) Viral Immunodiagnosis p. 125, Acad. Press.
3. Galardy, R.E., et.al. (1978) J. Med. Chem. 21, 1279.
Storage 0-4 °C.
Pyridine dithioethylamine hydrochloride
C5H11NO2S2; Mr=217.74; mp 128-129 °C
![]()
●
Sulfhydryl amination reagent.1
●
Used to conjugate Mitomycin C to Folic acid.2
●
Wide use in diagnostics, and conjugation chemistry.
Water soluble!
● Quantitate thiol groups by the release of 2-thiopyridone lmax=343
nm. e=8,080 M-1 cm-1.
1. Johnson, R.J., et.al. (1985) J. Biol. Chem.
260, 7161-7164.
2. Christopher P. Leamon, et.al. (2005) Bioconjugate Chem 16,
803-811.
Storage 0-4 °C.
1.0g
$400.00
[92944-71-3]; C17H11NO3; Mr=277.28; mp 155-157 °C
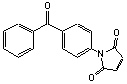
●
Sulfhydryl reactive photocrosslinking reagent.1,2
● Generates photoactivatable conjugates from thiols.3
● Photoactivation
occurs at 350 nm.
1. Agarwal, R., et.al. (1991) J. Biol. Chem. 266, 2272.
2. Leszyk, J., et.al. (1988) Biochemistry 27, 6983.
3. Tao, T., et.al. (1984) Biophys. J. 45, 261.
Storage 15-25 °C.
500mg
$275.00
C18H13NO5; Mr=323.30; mp 205-206 °C
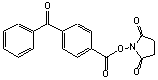
●
Amino reactive photocrosslinking reagent.1
●
Converts amines into photoaffinity probes or surface modification reagents.2
1. Rajasekharan, K.N., et.al. (1991) Arch. Biochem.
Biophys. 288, 584.
2. Parker, J.M.R., et.al. (1985) J. Protein Chem. 3, 479.
Storage 0-4 °C.
500mg
$275.00
C15H12NO2I; Mr=365.17
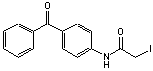
●
Sulfhydryl reactive photocrosslinking reagent.
● Useful for conjugating to carbohydrates,1 or mapping myosin structure.2
● Used to
crosslink chymotrypsin with unusually high efficiency (~100%).3
1. Hall, L.D., Yalpani, M. (1980) Carbohydrate Res. 78, C4.
2. Lu, R.C., Wong, A. (1989) Biochemistry 28, 4826.
3. Campbell, P., Gioannini, T.L. (1979) Photochem. Photobiol.
29, 883.
Storage 0-4 °C.
(N-Succinimidyl 3-[2-pyridyldithio]propionate)
[68181-17-9]; C12H12N2S2O4; Mr=312.40; mp 78-79 °C
Spacer Arm length: 6.8 Ĺ.
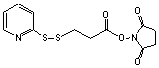
●
Sulfhydryl and amino reactive heterobifunctional protein
crosslinking reagent.1,2
● Protein thiolation reagent.1
● Tethering to artificial membranes.2
● Preparing antibody-toxin conjugates.3
● Hapten preparation.4
● Sulfhydryl modification can be followed by release of 2-thiopyridone, lmax=343
nm. e=8,080 M-1 cm-1.
1. Carlsson, J., et.al. (1978) Biochem. J. 173, 723-737.
2. Walden, P., et.al. (1986) J. Mol. Cell Immunol. 2, 191-197.
3. Cumber, A.J., et.al. (1985) Meth. Enzymol. 112, 207-225.
4. Gordon, R.D., (1978) PNAS 84, 308-312.
Storage 0-4 °C.
250mg
1.0g
$325.00
$995.00
(3-[2-Pyridyldithio]propionyl hydrazide)
C8H11N3S2O; Mr=229.32; mp 92-93 °C
Spacer Arm length: 9.2 Ĺ.
![]()
●
Sulfhydryl and carbohydrate reactive derivative of SPDP.
DTT or TCEP cleavable.
● Useful for preparing carbohydrate linked immunoconjugates.1
● Conjugating antibodies to nerve growth factor (NGF).2
1. Zara, J.J., et.al. (1991) Anal. Biochem. 194, 156-162.
2. Friden, P.M., (1993) Science 159, 373-378.
Storage 0-4 °C.
(Succinimidyl 6-[3-(2-pyridyldithio)propionamido]hexanoate)
C18H23N3S2O5; Mr=425.52; mp 82-84 °C
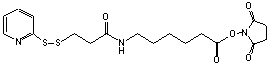
●
Sulfhydryl and amino reactive heterobifunctional protein
crosslinking reagent.
● Extended analog of SPDP with similar properties and uses.1
1. Carlsson, J., et.al. (1978) Biochem, J. 173, 723-737.
Storage 0-4 °C.
(sulfo-Succinimidyl 6-[3-(2-pyridyldithio)propionamido]hexanoate)
C18H22N3S3O8Na; Mr=527.56
Spacer Arm length: 15.6 Ĺ.
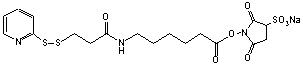
●
Sulfhydryl and amino reactive heterobifunctional protein
crosslinking reagent.
● Extended analog of SPDP with similar properties and uses1
●
Water soluble!
1. Carlsson, J., et.al. (1978) Biochem, J. 173, 723-737.
Storage 0-4 °C.
C16H19N3O6; Mr=349.34; mp 206-207 °C
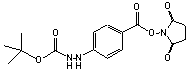
●
Amino reactive heterobifunctional crosslinking reagent with
latent hydrazino group.
●
Modification of chromatographic supports.1
● Hydrazino unmasked with TFA, HCl,
or formic acid.
1. Millington, C.R., et.al. (1998) Tet Lett. 39, 7201.
Storage 0-4 °C.
C11H17N3O6; Mr=287.27; mp 145-146 °C (Decomp)
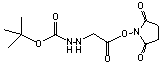
●
Amino reactive heterobifunctional crosslinking reagent with
latent hydrazino group.
● Hydrazino unmasked with TFA, HCl,
or formic acid.
Storage 0-4 °C.
2-Iminothiolane hydrochloride
[4781-83-3]; C4H8SNCl; Mr=137.63; mp 193-194 °C
![]()
●
Water soluble! Amino
reactive thiolation reagent.1,2,3
● An effective RNA-protein crosslinking reagent.4
● Thiolation reagent for polysaccharides.5
1. Traut, R., et.al. (1973) Biochemistry 12, 3266.
2. Jue, R., et.al. (1978) Biochemistry 17, 5399-5405.
3. Kenny, J.W., et.al. (1979) Meth. Enzymol. 59, 534.
4. Wower, L., et.al (1981) Nucleic Acids Res. 9, 4285.
5. Alagon, C.A., et.al. (1980) Biochemistry 19, 4341.
Storage 0-4 °C.
[6953-60-2]; C6H6SO4; Mr=174.18; mp 86-87 °C
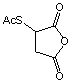
● Reacts with
primary amines to add protected sulfhydryls.1
● Thioacetyl group
is deprotected with 0.02 M hydroxylamine hydrochloride to release free
sulfhydryl.
1. Klotz, I.M.; Heiney, R.E. (1962) Arch.
Biochem. Biophys. 96, 605-612.
Storage 0-4 °C.
6-(N-trifluoroacetyl)caproic acid NHS
C12H15N2O5F3; Mr=324.25; mp 73-74 °C
Spacer Arm length: 7.7 Ĺ.

●
Amino reactive tether with protected amino group.
●
Amino group unmasked at pH 7.8-8.1, phosphate buffer (usually
25 °C/15-20 min.).
Storage 0-4 °C.
C15H24N2O6; Mr=328.36; mp 81-82 °C
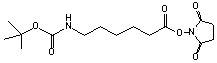
●
Amino reactive tether with protected amino group.
●
Amino group unmasked with formic acid (25
°C/20 min).
Storage 0-4 °C.
Succinimidyl-6-(iodoacetyl)aminocaproate
[134759-23-2]; C12H17N2O5I; Mr=396.18
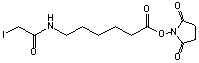
● Sulfhydryl and amino reactive heterobifunctional crosslinking reagent.1
1. Brinkley, M. (1992) Bioconjugate Chem. 3, 2.
Storage 0-4 °C.
Methyl N-succinimidyl adipate
C11H15NO6; Mr=257.24; mp 53 °C
Spacer Arm length: 7.2 Ĺ.
●
Amino reactive with latent carboxylate group.
● Carboxyl unmasked at pH 9.5, phosphate buffer.
Storage 0-4 °C.